|
MMM, something smells good. It smells like someone’s baking cookies! Even though you can’t see anyone making cookies, you know this by the familiar scent in the air. Tiny traces of molecules that give cookies their odor are floating through the air and detected by your nose. |
 |
 |
Think of other examples of things you’ve smelled without seeing. Hamburgers grilling on a BBQ. Freshly made popcorn. Perfume. These odors are familiar to you, and their concentration in the air is high enough for your nose to detect them. However, your nose isn't sensitive enough to detect everything in the air. There are many other things in the air in such small amounts that you would never be able to smell them. Other molecules, such as water vapor or carbon dioxide, are odorless. But that does not mean that they are not present in the air. Let's learn how scientists are able to detect all of these chemicals in the air. |
How do chemicals get in the air?
 |
Some chemicals are more likely to evaporate than others. This characteristic is called volatility (VOL-ah-TIL-i-tee). A chemical with high volatility is more likely to evaporate into a gas than a chemical with low volatility. What are some examples of chemicals with high volatility? Any type of fuel is extremely volatile. So much so that you can actually see the fumes above a fuel pump at a gas station.. You can also readily smell acetone as soon as a fingernail polish remover container is opened. What about other chemicals that have low volatility? Water, oil and aluminum are just a few examples. |
|
As gases, some volatile (VOL-ah-til) chemicals travel very high up into the atmosphere. When they reach high altitudes in the troposphere, approximately 5 to 9 miles above the earth’s surface, they can be transported very quickly over long distances. In some cases, it can take just days for a chemical to travel over oceans to distant continents or from one side of a continent to another. |
 |
How does this affect human health?
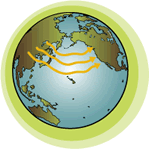 |
Some of the chemicals that are transported long distances in the atmosphere can be potentially harmful to people, wildlife, and plants. For that reason, it is extremely important to know where a chemical may go in the environment. Depending on its chemical properties, a chemical used in U.S. agriculture or industry could end up in the Alps or in the Arctic. Even trace amounts of some chemicals can cause harm in more sensitive environments. |
